Molecule of the Month: Apolipoprotein B-100 and LDL Receptor
Insights into how LDL is removed from the bloodstream
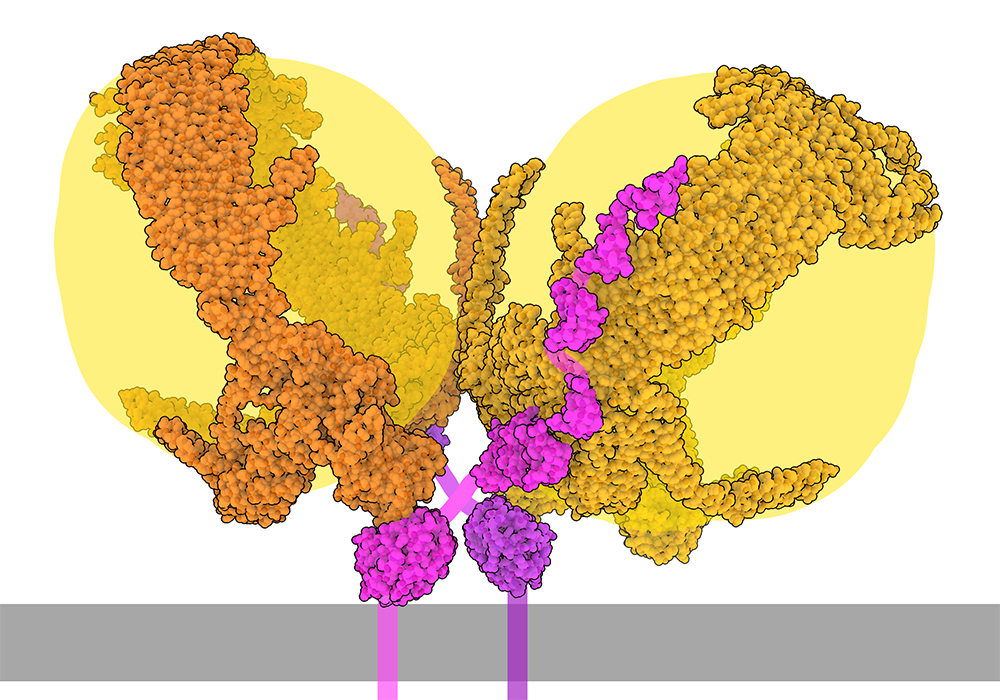
A Fat Delivery System
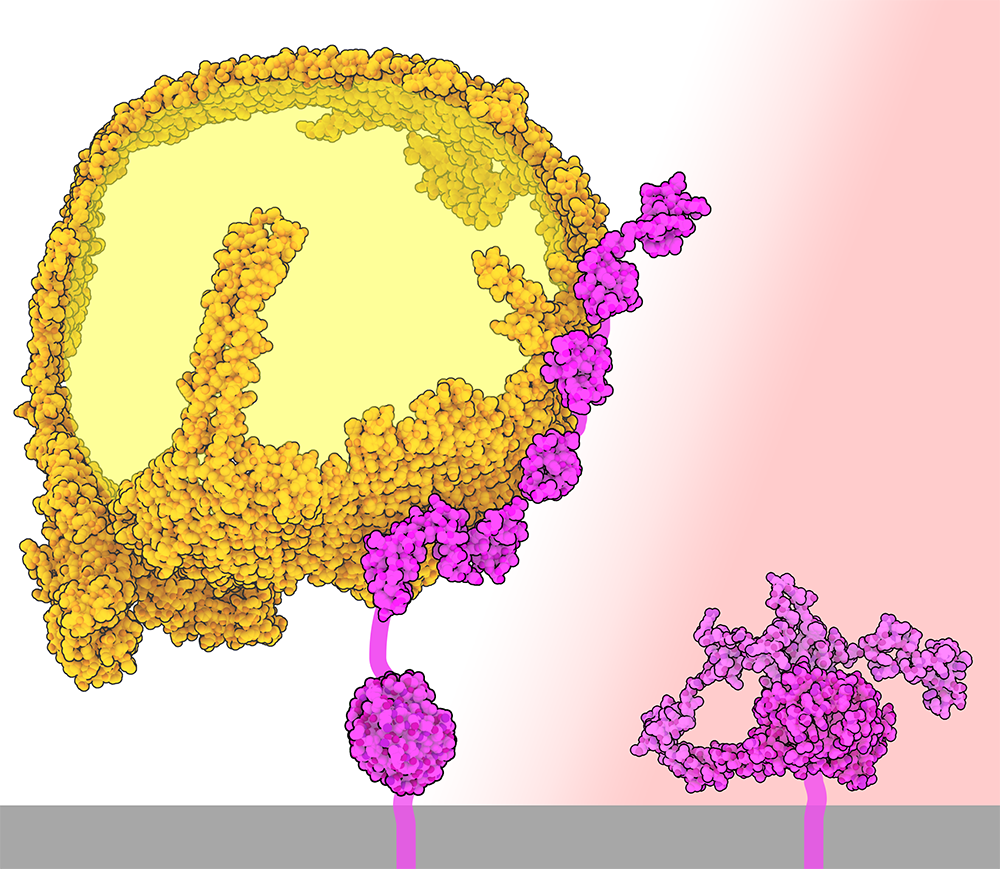
Letting Go of LDL
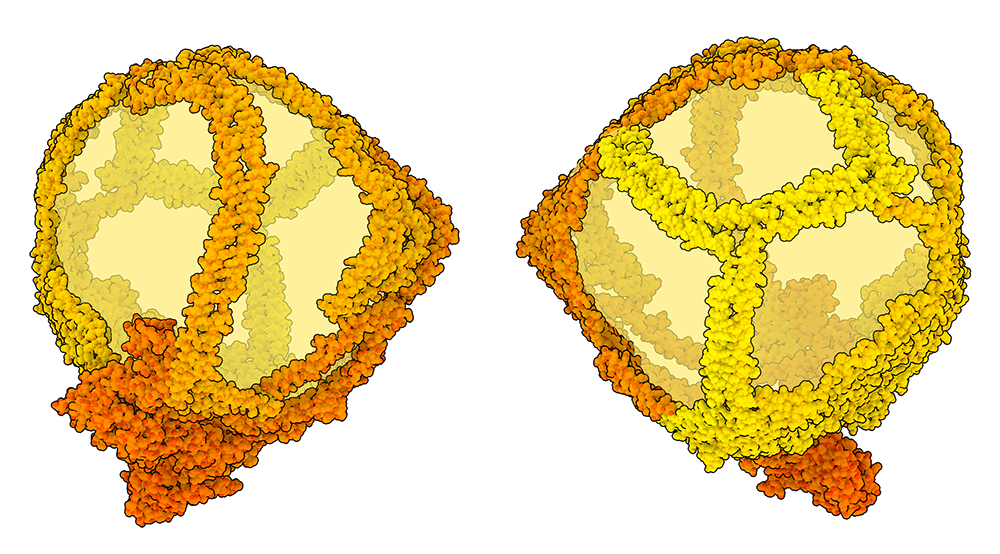
Adjusting for Size
Exploring the Structure
Mutations Causing Familial Hypercholesterolemia

If excess cholesterol is allowed to circulate in the bloodstream for an extended period of time, cholesterol-rich plaques can form in and on the walls of arteries, a disease known as atherosclerosis. Over time, arteries can become narrow or blocked, causing strokes or heart attacks. Familial hypercholesterolemia is a hereditary disorder that is characterized by high levels of circulating LDL. In many cases, this disease is caused by mutations in the genes encoding ApoB100 or the LDL receptor. You can take a closer look at mutations that have been localized to the binding interface between ApoB100 and LDL receptor by clicking on the JSmol tab.
Topics for Further Discussion
- Explore how apolipoproteins inspired the engineering of nanodiscs, a key tool for the structural study of membrane proteins.
- Take a closer look at how lipids are stored in lipid droplets in cells.
Related PDB-101 Resources
References
- 9BDT: Reimund M, Dearborn AD, Graziano G, Lei H, Ciancone AM, Kumar A, Holewinski R, Neufeld EB, O'Reilly FJ, Remaley AT, Marcotrigiano J. Structure of apolipoprotein B100 bound to the low-density lipoprotein receptor. Nature. 2024 Dec 11; 638:829-835.
- 1N7D: Rudenko G, Henry L, Henderson K, Ichtchenko K, Brown MS, Goldstein JL, Deisenhofer J. Structure of the LDL receptor extracellular domain at endosomal pH. Science. 2002 Dec 20;298(5602):2353-8.
- 9EAG: Berndsen ZT, Cassidy CK. The structure of apolipoprotein B100 from human low-density lipoprotein. Nature. 2024 Dec 11; 638:836-843.
April 2025, Janet Iwasa
http://doi.org/10.2210/rcsb_pdb/mom_2025_4


