SARS-CoV-2 mRNA Vaccine
Atomic structures are being used to design effective vaccines against SARS-CoV-2

|
| Messenger RNA (mRNA) vaccines developed for the COVID-19 pandemic are composed of long strands of RNA (magenta) that encode the SARS-CoV-2 spike surface glycoprotein enclosed in a lipid membrane (blue) that delivers the RNA into cells. The membrane surface is decorated with polyethylene glycol chains (green) that help shield the vaccine from the immune system, lengthening its lifetime following administration. In this illustration, the vaccine is shown surrounded by blood plasma. A high-resolution file for this illustration is available in the SciArt Gallery. |
Vaccines represent our best hope for controlling the SARS-CoV-2 pandemic. It’s currently thought that about 60%-70% of us would need to have immunity in order for us to be safe to return to normal social activities. Unfortunately we can’t rely on natural immunity for this, since it would lead to tens of millions of deaths worldwide (millions alone in the US), given the fatality rate of this virus, particularly in older individuals. Vaccines provide the safest path to immunity, and to this end, researchers around the globe are trying every imaginable approach to creating a vaccine.
Harnessing our Own Cells
Vaccines typically work by presenting our immune system with pieces of a virus, or even weakened versions of a whole virus. The immune system then builds up defenses to fight these pieces. These defenses protect us when we’re challenged by a real virus. The new mRNA vaccines harness our own cells to build these pieces of viruses. The heart of the vaccine is a strand of RNA that encodes the spike surface glycoprotein of SARS-CoV-2. The outer envelope of the vaccine is designed to deliver this RNA into cells, where it directs construction of many copies of the spike protein. The cell displays these proteins on its surface, where they mobilize the immune system to mount a protective response.
|
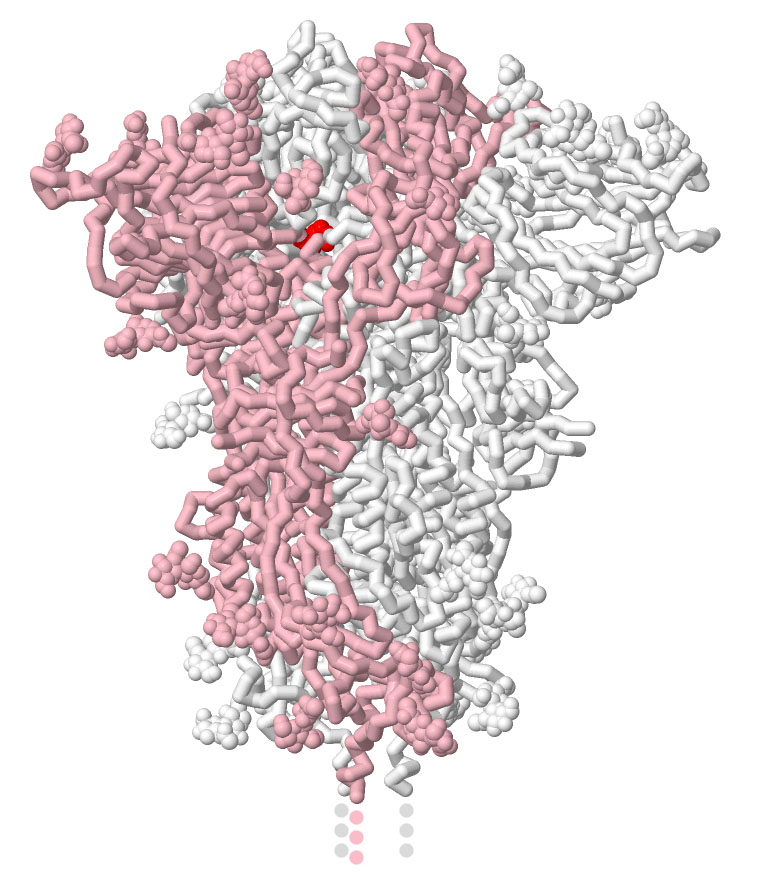
| SARS-CoV-2 spike with mutations at two neighboring sites that stabilize the closed form. One chain of the trimeric spike is shown in pink with the mutations in red. |
Specialized Spike
Researchers are doing everything they can to ensure that the strongest possible immune response is elicited by the vaccine. One way they do this is to stabilize the spike protein in a conformation that is highly immunogenic. The spike protein has evolved to undergo many structural changes, some of which are undesirable for vaccination purposes. So, researchers have incorporated targeted mutations to freeze it into the shape that it adopts when people first encounter the virus. The mutated spike structure shown here (PDB entry 7jji) stabilizes a sharp turn in the polypeptide chain by changing two consecutive amino acids to prolines, which favors the closed form of the spike. Using the growing body of structural information available in the PDB archive, researchers are also trying out many other modifications, including changes to the cleavage site that activates the spike, and addition of disulfide linkages to glue the entire protein together.
|
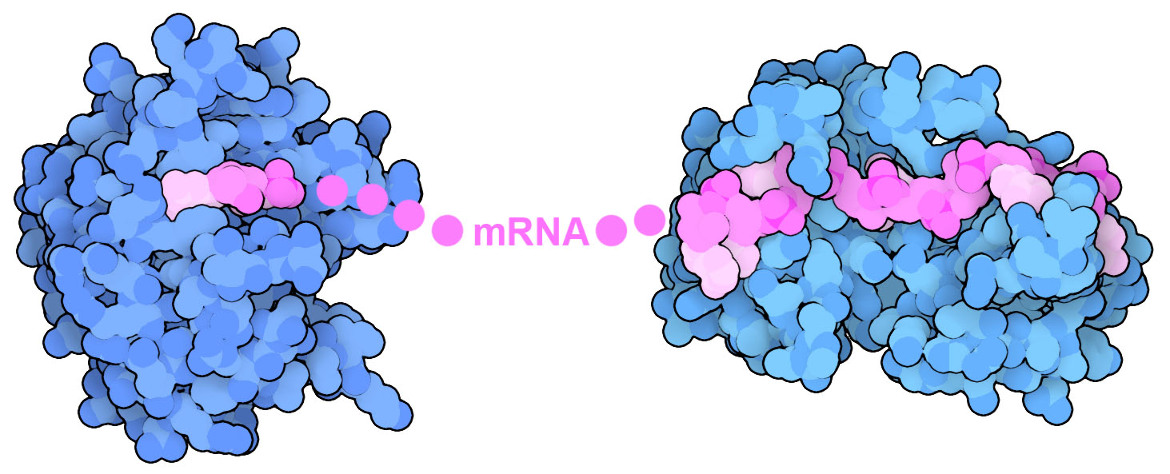
| Structures of messenger RNA 5’ cap-binding protein EIF4E (left) and poly(A)-binding protein (right) show how the two ends of mRNA are recognized. |
Engineering the Message
The mRNA carried inside the vaccine is also highly optimized to maximize effectiveness. It needs to work exactly like a normal cellular mRNA when the cell’s own protein translation system builds the immunogenic spike protein. So, the vaccine mRNA is constructed with a typical cap at the front end, composed of a modified guanine nucleotide connected backwards to the chain, and a long tail of repeated adenine nucleotides at the back end. Inside cells, these two features are recognized by specialized proteins, shown here from PDB entries 1cvj and 1ej1. Not-so-typical changes in the mRNA are also made, such as using a slightly-modified form of uracil to help the vaccine elude the innate immune defenses that help protect us from viral RNA. Together, these modifications help ensure that the mRNA is processed normally inside a cell, where it can begin its work stimulating the immune system.
More Information
- The Nobel Prize in Physiology or Medicine 2023: Katalin Karikó and Drew Weissman “for their discoveries concerning nucleoside base modifications that enabled the development of effective mRNA vaccines against COVID-19”
- Corbett, K. S., et al. (2020) SARS-CoV-2 mRNA vaccine designs enable by prototype pathogen preparedness. Nature 586, 567-571.
- Nelson, J. (2020) Impact of mRNA chemistry and manufacturing process on innate immune activation. Science Advances 6, eaaz6893.
- Vogel, A. B., et al. (2020) A prefusion SARS-CoV-2 spike RNA vaccine is highly immunogenic and 1 prevents lung infection in non-human primates. bioRxiv preprint doi: https://doi.org/10.1101/2020.09.08.280818
Entries from the PDB Archive Discussed in this Article
- 7JJI: Bangaru, S. (2020) Structural analysis of full-length SARS-CoV-2 spike protein from an advanced vaccine candidate. Science 370, 1089-1094.
- 1CVJ: Deo, R. C., et al. (1999) Recognition of polyadenylate RNA by the poly(A)-binding protein. Cell 98, 835-845.
- 1EJ1: Marcotrigiano, J., et el. (1997) Cocrystal structure of the messenger RNA 5’ cap-binding protein (eIF4E) bound to 7-methyl-GTP. Cell 89, 951-961.



