Myohemerythrin
1985, 13 7/8" x 10 7/8"

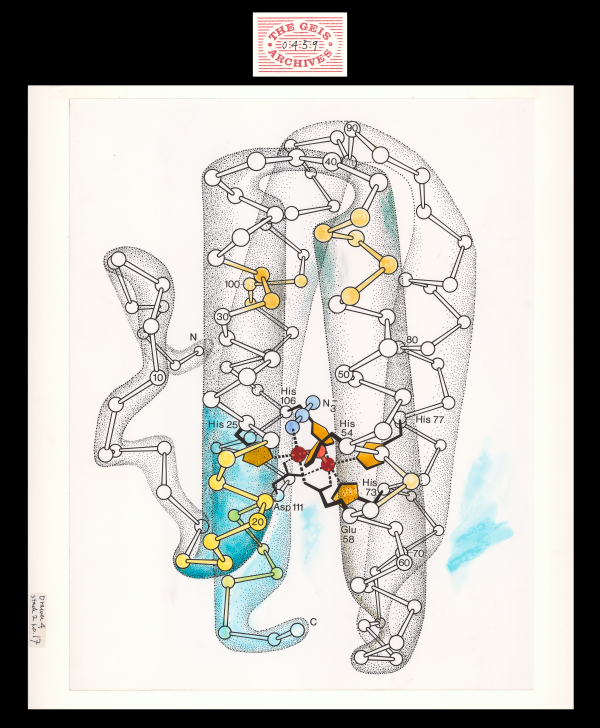
The colored print depicts the structure of myohemerythrin, which was first revealed by X-ray crystallography in 1975 (Hendrickson et al., 1975) and further refined in 1987 (Sheriff et al., 1987). Geis's illustration depicts the tertiary structure of the protein, highlighting the four anti-parallel alpha-helices and the presence of mu-oxo-diiron (iron atoms in red and oxygen atom in pink) located within the core of the macromolecule (Myohemerythrin).
Used with permission from the Howard Hughes Medical Institute (www.hhmi.org). All rights reserved.

Related PDB Entry: 2MHR
Experimental Structure Citation
Sheriff, S., Hendrickson, W. A., & Smith, J. L. (1987). Structure of myohemerythrin in the azidomet state at 1.7/1.3 Å resolution. J. Mol. Biol. 197, 273-296.
About Myohemerythrin
Myohemerythrin, like myoglobin, stores oxygen in the muscles of many marine invertebrates ("Myohemerythrin" website). The two iron atoms are responsible for binding oxygen. One iron atom (shown in red in static rendition) is coordinated by two histidine residues (His25 and His54) (orange and grey in static rendition) and the other iron atom (red in static rendition) is coordinated by three histidine residues (His73, His77, and His106) (orange and grey in static rendition). The iron atoms are bridged by glutamic (Glu58) and aspartic acid (Asp111) sidechains (grey in static rendition). They are also bridged by oxide (O2-) (pink in static rendition). Oxygen is capable of binding to azide (N3-) (blue in static rendition) as hydroperoxide (HO22-). The four helix bundle is stabilized by van der Waals interactions among buried hydrophobic residues.
Text References
Myohemerythrin. University of Wisconsin - Eau Claire, Department of Chemistry, Interactive Biochemistry. URL: http://www.chem.uwec.edu/Modeling/GGProteinStructures/Pages/RPS/myohemerythrin/myohemerythrin.html.
Initial Structure Determination References
Hendrickson, W. A., Klippenstein, G. L., & Ward, K. B. (1975). Tertiary structure of myohemerythrin at low resolution. Proc. Natl. Acad. Sci. USA, 72, 2160-2164.