Molecule of the Month: Epidermal Growth Factor
EGF is part of a family of proteins that controls aspects of cell growth and development

Introduction
Domains and Dimers
Structural Surprises
Large Family

Turning the Receptor Off
EGF Receptor Kinase Domain with Lapatinib (PDB entry 1xkk)

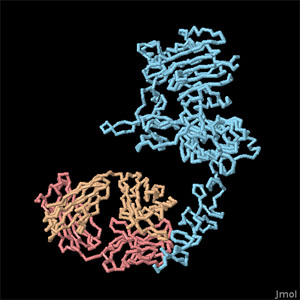
The signal carried by EGF can be dangerous if used improperly. Many forms of cancer circumvent the normal EGF signaling process, giving themselves permission to grow without control. Because of this, drugs that block EGF signaling are effective for the treatment of cancer. Two examples are shown here. At the left, the drug lapatinib is bound in the kinase domain of the receptor, blocking the signal inside the cell (PDB entry 1xkk ). It is very similar to the ATP used by the receptor, and binds tightly in the active site. Therapeutic antibodies are also used for cancer treatment. Herceptin is shown on the right bound to the extracellular domain of HER2/ErbB2 (PDB entry 1n8z), and the antibody cetuximab bound to the EGF receptor may be found in PDB entry 1yy9. To explore these structures in more detail, click on the images for an interactive Jmol image.
Topics for Further Discussion
- Compare the mode of dimerization in the EGF receptor and the
- human growth hormone receptor.
- EGF receptor and similar receptors are currently the targets of development of drugs
- for cancer therapy. Can you find other examples of structures in the PDB with drugs?
- Compare the mode of dimerization in the EGF receptor and the
- human growth hormone receptor.
- EGF receptor and similar receptors are currently the targets of development of drugs
- for cancer therapy. Can you find other examples of structures in the PDB with drugs?
Related PDB-101 Resources
- Browse Cellular Signaling
- Browse Cancer
References
- M. Lemmon (2009) Ligand-induced ErbB receptor dimerization. Experimental Cell Research 315, 638-648.
- R. Bose and X. Zhang (2009) The ErbB kinase domain: structural perspectives into kinase activation and inhibition. Experimental Cell Research 315, 649-658.
- K. M. Ferguson (2008) Structure-based view of epidermal growth factor receptor regulation. Annual Review of Biophysics 37, 353-373.
June 2010, David Goodsell
http://doi.org/10.2210/rcsb_pdb/mom_2010_6


