Molecule of the Month: Ras Protein
Mutation of the growth-controlling ras protein can lead to cancer
Introduction
Ras Oncogene
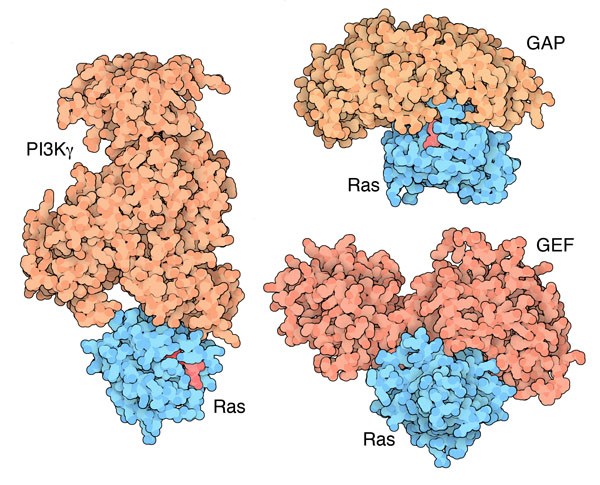
Ras Partners
Exploring the Structure
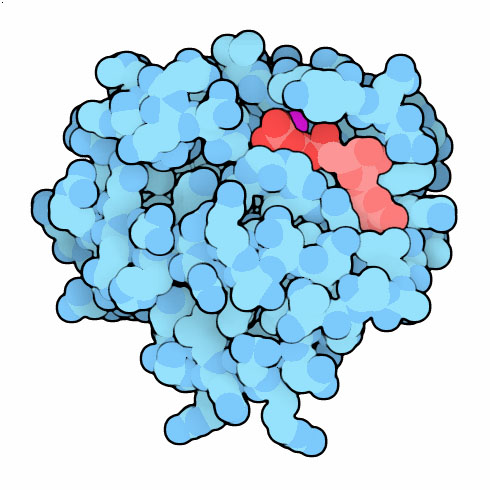
Ras Protein

Many structures for Ras have been solved, showing the different conformational states in the signaling cycle. The two shown here capture Ras with GTP and GDP, in the on and off states (PDB entries 5p21 and 4q21 ). Comparison of these structures revealed that two loops (in green) near the nucleotide change conformation. This is the signal that is recognized by the effector proteins that transmit the signal. Structures have also revealed the importance of glutamine 61, which positions a water molecule to perform the cleavage reaction. This glutamine is often mutated in cancer cells, so the GTP is never cleaved and the protein is always turned on. To see several other structures that show the action of this amino acid, click on the image for an interactive Jmol.
Topics for Further Discussion
- Structures for several mutants of Ras have been solved, including Ras with mutations of glutamine 61. You can use the Structure Comparison tool to compare these to the normal form of the protein.
- Many structures of Ras have been solved with modified forms of GTP, with nitrogen or carbon atoms connecting the phosphates. Why is this necessary?
Related PDB-101 Resources
- Browse Cancer
- Browse Cellular Signaling
References
- Y. Pylayeva-Gupta, E. Grabocka and D. Bar-Sagi (2011) Ras oncogenes: weaving a tumorigenic web. Nature Reviews Cancer 11, 761-774
- I. R. Vetter and A. Wittinghofer (2001) The guanine nucleotide-binding switch in three dimensions. Science 294, 1299-1304.
April 2012, David Goodsell
http://doi.org/10.2210/rcsb_pdb/mom_2012_4


