Molecule of the Month: Crystallins
A concentrated solution of crystallins refracts light in our eye lens
Crystal Clear
Transparency Through Diversity
Moonlighting Proteins
Cataracts
Exploring the Structure
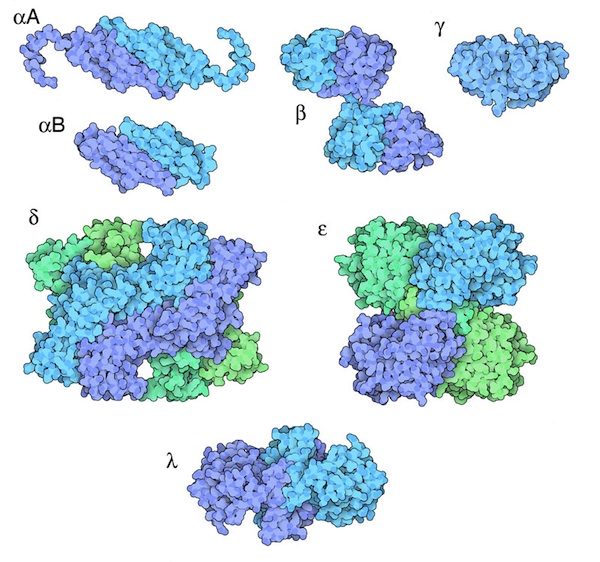
Beta and Gamma Crystallin (PDB entries 1blb and 4gcr)

The key to crystallin function is their ability to form many different types of similar complexes, so that they will form a smooth, random arrangement when concentrated inside lens cells. The crystal structures of crystallins have revealed that they use domain swapping to form many different complexes using a small number of building blocks. Beta crystallin uses a classic domain swapping mechanism to mix-and-match different variants of the beta chain, and alpha crystallins use a more complex mechanism to create larger complexes. Click the images above to see Jmols that show these domain swaps in detail.
Topics for Further Discussion
- Many proteins use domain swapping to create stable complexes. Can you find other examples in the PDB? (Hint: many researchers mention the term "domain swapping" in their titles or abstracts).
- Can you think of any other proteins with two or more different functions?
Related PDB-101 Resources
- Browse You and Your Health
- Browse Biomolecules
References
- K. K. Sharma and P. Santhoshkumar (2009) Lens aging: effects of crystallins. Biochimica et Biophysica Acta 1790, 1095-1108.
- L. Takemoto and C. M. Sorensen (2008) Protein-protein interactions and lens transparency. Experimental Eye Research 87, 496-501.
- H. Bloemendal, W. de Jong, R. Jaenicke, N. H. Lubsen, C. Slingsby and A. Tardieu (2004) Ageing and vision: structure, stability and function of lens crystallins. Progress in Biophysics and Molecular Biology 86, 407-485.
July 2010, David Goodsell
http://doi.org/10.2210/rcsb_pdb/mom_2010_7


